Therapeutic area: Metabolic Disorders
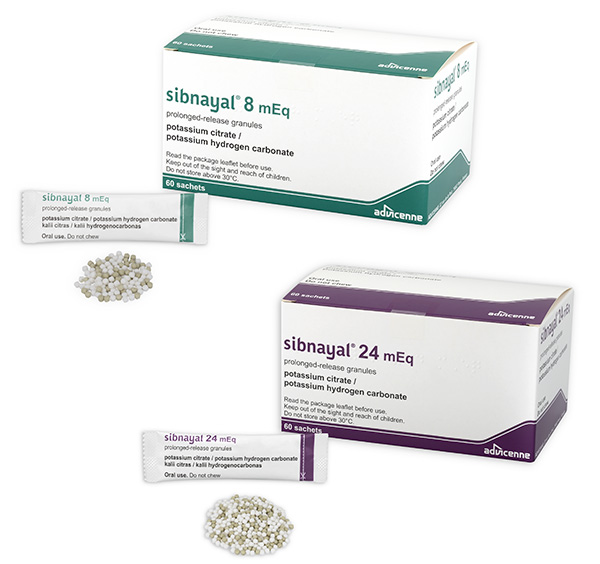
Sibnayal® :
Active substances : potassium citrate and potassium hydrogen carbonate
Dosages :
- Sibnayal 8 mEq, prolonged-release granules
- Sibnayal 24 mEq, prolonged-release granules
Conditions of supply and use :
- Medicinal product subject to medical prescription.
SIBNAYAL® has been approved by EMA in April 30th, 2021.
For more information about Sibnayal® please use the link below:
Advicenne signed partnerships with 3 companies for the commercialisation of Sibnayal® in Europe : FrostPharma AB for the Nordic Region of Europe, TwinPharma for Benelux and ExCEEd Orphan for Central and Eastern Europe.